|
If the DNA insert is generated from restriction enzyme digestion, the 5'-phosphate group is already present. If the DNA inserts you are ligating have blunt ends, the inserts must be 5'-phosphorylated at both ends in order for ligation to occur. To further help prevent vector re-circulization, treating the vector with DNA phosphatase helps to remove the 5’-end phosphate groups from the vector before the ligation step. Using a higher DNA insert to vector ratio is recommended to help ensure ligation of your DNA insert into your vector while preventing vector re-circulization (ligation of your vector without the DNA insert). Since blunt-end ligation is less efficient than sticky end ligation, this approach will require additional optimization and planning. For this approach, you can either choose a restriction enzyme that will generate blunt ends, or you can generate sticky ends and remove the overhangs by using an end repair kit. For those cases, you would choose a blunt-end cloning strategy to ligate your DNA insert into your vector as illustrated in the fourth panel of Figure 2. However, it is not always possible to use restriction enzymes that cut in different places to create sticky ends. When compared with the alternative (blunt-end ligation), sticky end ligation is more efficient due to the compatible overhangs that assist with the ligase reaction. 
Another reason for using sticky ends is to increase your ligation efficiency. For example, if you want to clone your DNA insert so that it is read in a specific direction or orientation (also known as directional cloning), sticky end ligation is the method of choice since the created overhangs will only ligate in a specific orientation. These “sticky ends” can be very helpful and are something that should be considered first when designing your cloning strategy. These fragments are created when restriction enzymes cut in different places within the double-stranded DNA, resulting in overhangs (unpaired nucleotides). The first three panels in Figure 2 illustrate strategies for cloning fragments with distinct sticky ends. T4 DNA ligase activity requires Mg2+ and ATP to work, and requires 5'-phosphorylation of one or both fragments. T4 DNA ligase can catalyze a reaction between blunt-end (no overhangs) or sticky end (3' or 5' complementary single-stranded overhangs) DNA fragments. T4 DNA ligase is an enzyme that helps create the formation of a phosphodiester bond between the 3'-hydroxyl end of a double-stranded DNA fragment and the 5'-phosphate end of the same or another DNA fragment (Figure 1). This is typically done by using T4 DNA ligase.  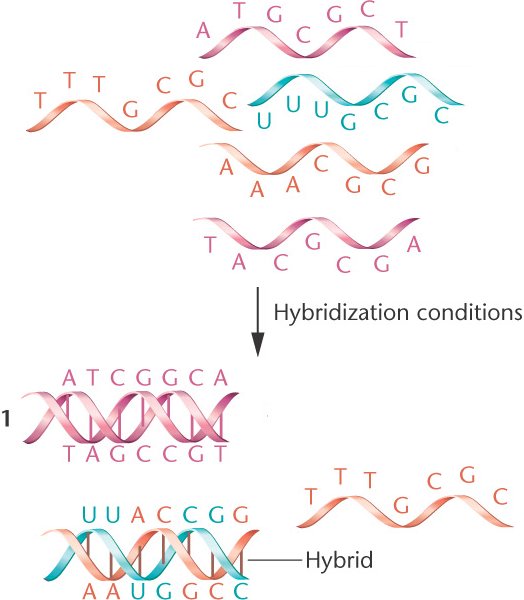
Ligation works by using a phosphodiester bond to connect the sugar backbone of the double-stranded DNA insert with the sugar backbone of the double-stranded DNA vector. The final step, ligation (aka the pasting step) is used to seal the insert into the vector. This process involves multiple steps (such as copying the DNA, cutting out the gene of interest, and pasting the gene into the DNA vector). Molecular cloning is the process used for taking recombinant DNA (referred to as an insert) and placing it into a DNA vector (i.e., plasmid) where it can be replicated and expressed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
